Nov 6 2014
Photosynthesis is one of the most important processes in nature. The complex method by which all green plants harvest sunlight and thereby produce the oxygen in our air is still not fully understood. Researchers have used DESY's X-ray light source PETRA III to investigate a photosynthesis subsystem in a near-natural state.
According to the scientists led by Privatdozentin Dr. Athina Zouni from the Humboldt University (HU) Berlin, X-ray diffraction experiments on the so-called photosystem II revealed structures which were yet unknown. The results are published in the scientific journal Structure. The method applied might also be of interest for the structural analysis of other biomolecules.
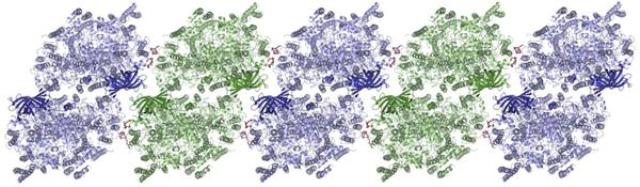 Molecular structure of photosystem II, which arranges itself in rows. Credit: Martin Bommer/HU Berlin
Molecular structure of photosystem II, which arranges itself in rows. Credit: Martin Bommer/HU Berlin
Photosystem II is the part of the photosynthetic machinery, which splits water into hydrogen and oxygen with the help of sunlight. It is located in the cellular membrane. Such membrane proteins are a large and vital group of biomolecules that are also important in addressing a variety of medical issues. In order to determine the structure of a protein and reveal details on its biomolecular function, researchers use the very bright short-wave X-rays of PETRA III and similar facilities. For such experiments single crystals must be grown from the biomolecules. “The structure of single molecules cannot be observed directly, even with the brightest X-rays,” explains Dr. Anja Burkhardt, co-author and DESY researcher at the experimental station P11, where the measurements were carried out. “In a crystal, plenty of the same molecules are arranged in a highly symmetrical fashion. Thus the signal, resulting from X-ray diffraction of these molecules, is amplified. The molecular structure can then be calculated from the collected diffraction images.”
Biomolecules – and especially membrane proteins – do not easily adopt a crystalline state because it contradicts their natural role inside the flexible cellular membrane. Preparing suitable samples is therefore a crucial step in structural analysis. Photosystem II must first be separated from the membrane, where it is bound to numerous lipid molecules. For this purpose, researchers use special detergents similar to those found in soap. But instead of being embedded in lipids, the biomolecules are now surrounded by detergents, which make the crystals spongy, and thereby deteriorate the analytical result. “Our goal is to get as close to the natural state as possible,” stresses Zouni, because the higher the similarity between the protein in the crystal and its natural state, the more meaningful are the results.
The group led by Zouni has now managed to produce photosystem II crystals, which no longer contain detergents so that the biomolecules can be analyzed in a near-natural state. “The trick was to use a detergent that is completely different from the lipids regarding its composition and structure,” explains the Berlin-based researcher. X-ray diffraction measurements are usually performed on frozen crystals because the radiation damage that high-energy X-rays induce in crystalline samples can be significantly reduced by data collection at low temperatures. During the freezing procedure, the researchers want to avoid ice formation. Thus, the water in macromolecular crystals is partially extracted and replaced by a cryo-protecting agent before the crystals are exposed to X-rays.
“The dehydration process removed not only the water in our samples, but also completely removed the detergent, something we didn’t expect,” says Zouni. “Our samples are closer to the natural state than previously reported structures.”
Consequently, the spatial resolution could be increased from about 0.6 nanometres (a nanometre is a millionth of a millimetre) to 0.244 nanometres. While this is not even the highest resolution ever achieved in a photosystem II study, the structural analysis shows that photosystem II proteins are arranged within the crystals as extended rows, something that also occurs in the natural environment.
Electron microscopy investigations by Professor Egbert Boekema’s group at the University of Groningen in the Netherlands had already shown the photosystems’ crystal-like arrangement in the natural membrane — a kind of extremely tiny solar cell. Electron microscopy was able to visualize the intermolecular alignment through direct observation of the native membrane, while X-ray crystallography now fills in the smallest structural details. “We superimposed the X-ray diffraction data with the electron microscopy images – they matched perfectly,” says Zouni. The investigation also revealed structures that were previously invisible. “We can see exactly where and how the lipids are bound,” the scientist explains. The more the researchers discover about photosystem II, the better they understand exactly how it functions.
Careful manipulation of the detergent system is not only interesting in the context of photosystem II. “This can potentially be applied to many other membrane proteins,” stresses Zouni. In the future, many biomolecules could maybe studied in a more natural state than ever before.
Reference:
"Native-like Photosystem II Superstructure at 2.44 A Resolution through Detergent Extraction from the Protein Crystal"; Julia Hellmich, Martin Bommer et al.; Structure, 2014; DOI: 10.1016/j.str.2014.09.007