Purdue University researchers are creating and testing patent-pending poly (lactic-co-glycolic acid), or PLGA, nanoparticles modified with adenosine triphosphate, or ATP, to improve immunotherapy against malignant tumors.
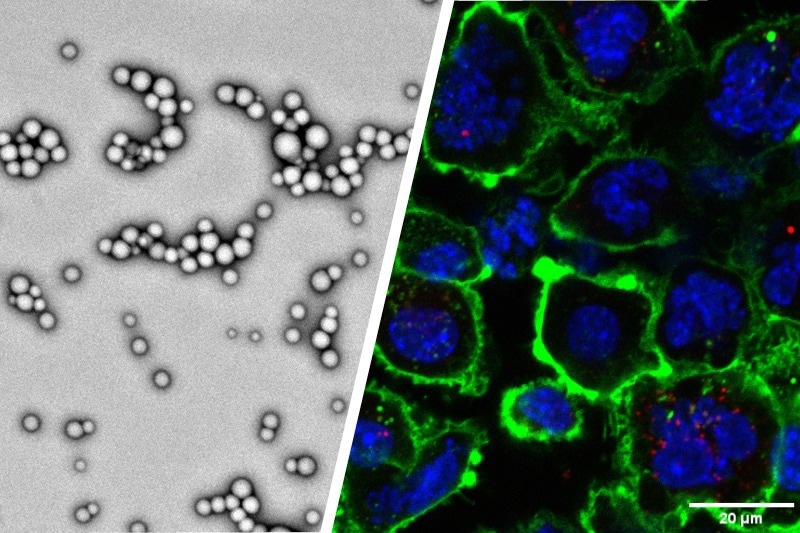 Purdue University researchers are developing and validating patent-pending nanoparticles (left) to enhance immunotherapy effects against tumors. The nanoparticles are modified with adenosine triphosphate, or ATP, to recruit dendritic cells (right), which are immune cells that recognize tumor antigens and bring specialized immune cells to fight off tumors. Image Credit: Yoon Yeo
Purdue University researchers are developing and validating patent-pending nanoparticles (left) to enhance immunotherapy effects against tumors. The nanoparticles are modified with adenosine triphosphate, or ATP, to recruit dendritic cells (right), which are immune cells that recognize tumor antigens and bring specialized immune cells to fight off tumors. Image Credit: Yoon Yeo
The nanoparticles release slowly drugs that cause immunogenic cell death (ICD) in cancers. ICD produces tumor antigens and other molecules that attract immune cells to a tumor’s microenvironment. The researchers added ATP to the nanoparticles, drawing immune cells to the tumor, triggering anti-tumor immune responses.
Yoon Yeo directs a team of researchers from the College of Pharmacy, the Metabolite Profiling Facility at the Bindley Bioscience Center, and the Purdue Institute for Cancer Research in the development of the nanoparticles.
Yeo is the associate department head and Lillian Barboul Thomas Professor of Industrial and Molecular Pharmaceutics and Biomedical Engineering, as well as a member of the Purdue Institute for Drug Discovery and the Purdue Institute for Cancer Research.
The researchers verified their findings with paclitaxel, a chemotherapy drug used to treat a variety of tumors. They discovered that tumors developed more slowly in mice treated with paclitaxel encapsulated in ATP-modified nanoparticles than in mice treated with paclitaxel in non-modified nanoparticles.
When combined with an existing immunotherapy drug, the ATP-modified, paclitaxel-loaded nanoparticles eliminated tumors in mice and protected them from rechallenge with tumor cells.
Yoon Yeo, Associate Department Head, Industrial and Molecular Pharmaceutics, Purdue University
ACS Nano published the study.
Challenges to Systemic Immunotherapy Delivery
Immunotherapy is a promising method for cancer treatment, but Yeo believes it does not benefit a huge number of people because they lack the robust immune cells required to assault tumors.
Yeo stated, “Pharmacological agents to activate immune cells can directly be given to tumors. Then the immune system can fight not only the treated tumors but also nontreated tumors in distant locations as the activated immune cells circulate in the bloodstream.”
However, Yeo stated that most tumors with a bad prognosis are not always easy to find or access. As a result, they may not respond well to local treatment. She and her colleagues envisioned systemic immunotherapy delivery, but there were hurdles.
“For successful systemic administration, active ingredients that stimulate anti-tumor immune responses need to be simultaneously present in tumors to exert concerted effects on the target. The ingredients also must maintain their activity until they reach tumors, but not cause toxic off-target effects. Moreover, the carriers traditionally used in local drug delivery offer limited utility in systemic application because they may not be compatible with blood components,” Yeo further added.
Yeo and her colleagues delivered immunotherapy compounds via biocompatible polymeric nanoparticles tailored to safely stimulate the immune system.
Yeo added, “We employed poly (lactic-co-glycolic acid), or PLGA, nanoparticles based on the strong track record of the polymer in FDA-approved products and its routine use in the systemic delivery of poorly water-soluble drugs.”
After several systemic administrations, ATP-modified PLGA nanoparticles were well tolerated in mice. They could mobilize dendritic cells, immune cells that detect tumor antigens and bring in specialist immune cells to combat cancers.
Yeo noted, “Moreover, the nanoparticles were shown to control the release of paclitaxel to minimize its systemic toxicity.”
The Next Development Steps
Yeo and her associates intend to conduct more research on the ATP-modified nanoparticles.
“We are currently working on improving the delivery of the nanoparticles to tumors and combining them with other treatments that will circumvent the resistance to the nanoparticle-delivered immunotherapy. To finance these efforts, we will apply for continued support from the National Institutes of Health. We are also open to industry partnerships to take this technology to the clinic,” Yeo added.
After Yeo revealed the nanoparticle idea to the Purdue Innovates Office of Technology Commercialization, the latter filed a patent application with the U.S. Patent and Trademark Office to safeguard intellectual property. Joe Kasper, assistant director of business development and licensing for biosciences, can be contacted at [email protected] regarding track code 69546 by industry partners who are interested in developing the compound or commercializing it for the market.
The Purdue Institute for Cancer Research, the Indiana Clinical and Translational Sciences Institute, the National Center for Advancing Translational Sciences, and the National Institutes of Health provided funding for Yeo's study.
Journal Reference:
Kwon, S., et. al. (2024) Systemic Delivery of Paclitaxel by Find-Me Nanoparticles Activates Antitumor Immunity and Eliminates Tumors. ACS Nano. doi:10.1021/acsnano.3c11445